Technology
RED - Blue Energy - Basics
REDstack is working intensively and successfully on the development, scaling up and future commercialisation of energy generation from differences in salinity, often referred to as Salinity Gradient Power (SGP). This form of energy - often called Blue Energy - was first described in 1954 by the English researcher Pattle in the journal Nature. Of course, the principle had to be translated into actual process technology and equipment that is technically sound, resistant, and stays in one piece, and is accepted by users in terms of price. This is the source of REDstack's mission.

The basis of SGP is laid down in the laws of thermodynamics. These say that a system strives for minimum energy and maximum entropy. The pursuit of minimal energy is seen in the normal world as apples falling from the tree. Entropy is sometimes referred to as 'disorder': particles tend to spread out over as large a volume as possible. In itself, ordinary mixing of fresh and salt water hardly releases any heat; it is mainly the entropy that increases dramatically. The salt particles of seawater can be compared to a gas trapped under a pressure of 25 bar (atmosphere). The salt, with the strength of a gas, wants to expand and electrical energy can be generated from it, but this requires technical facilities.
In order to be able to use this salt expansion for energy generation, this mixing process will have to take place in a controlled manner. In principle, two options are available for this. The first option is to install a membrane between salt and fresh water that only allows water through and blocks salt. Mixing can now only take place through water transport from fresh to salt. This phenomenon is called osmosis. A pressure of 25 bar can develop in the salt water compartment. The pressure of the salt water increases and can be used to drive a turbine. This technique is called Pressure Retarded Osmosis (PRO).
The second method for extracting this energy - and this is the method developed by REDstack, is to use membranes that allow only the charged salt particles (ions) to pass through. Salt ions, (Cl- and Na+), are very small, so that - given the right composition of the membrane - a fast, efficient and safe (no damage to the membrane) process can take place. This is the principle method that Pattle described in its essence. This is also the technique used by REDstack - now at the Afsluitdijk development site and soon in its commercial operating phase.
Information film
Blue is the new green.
Reverse ElectroDialysis (RED) - How it works
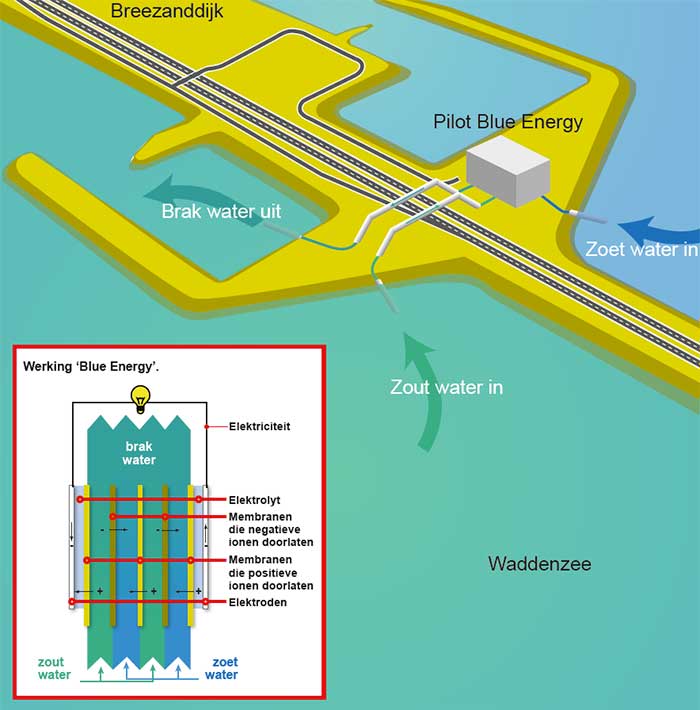
RED (Reverse ElectroDialysis, omgekeerde elektrodialyse) maakt gebruik van het feit dat zouten zijn opgebouwd uit ionen: positief en negatief geladen deeltjes. In zeewater zijn dat voornamelijk Na+ (het natrium-ion) en Cl- (het chloride-ion). En wanneer een zout opgelost is in water, komen deze ionen als losse deeltjes voor in het water. Bij RED worden nu twee type membranen gebruikt: membranen die uitsluitend positieve ionen doorlaten (CEM, Cation Exchange Membranes) en membranen die enkel negatieve ionen doorlaten (AEM, Anion Exchange Membranes).
When salt water flows between two such membranes (with fresh water on the other side of the membranes), the ions from the salt water will want to migrate to the fresh water. However, because of the membranes used, a CEM on one side and an AEM on the other, the two types of ions will therefore migrate in opposite directions. This creates a transport of positively charged parts in one direction and negatively charged parts in the other direction. So this creates a + and a - side, a kind of battery, an electrochemical cell.
The amount of electrical energy available is determined by the difference in salinity between the two water types, and the temperature of the water. The actual power generated depends on a number of technical issues such as the selectivity of the membranes used and the resistance of the electrochemical cell. The selectivity is the degree to which a membrane is able to pass only one ion species. And the resistance describes the hindrance that the ions experience on their way from the salt water to the fresh water.
Value of RED technology
Fossil fuels - coal, oil and natural gas - are still the main sources of energy today. These fuels have two major drawbacks: they are limited in supply and their combustion produces the greenhouse gas CO2. Because the easily accessible reserves are gradually being exhausted, people have been looking for new sources for a long time. We know these as shale gas and tar sand oil. The exploitation of these new sources, however, is accompanied by an ever-increasing assault on the environment, on nature and on our living space. In addition, the resulting CO2 is perhaps an even greater threat. Carbon dioxide forms an insulating blanket around the earth which results in the warming of the atmosphere with a consequent change in climate and a rise in the sea level. In time (over a thousand years) the amount of carbon dioxide may even become a public health problem. A third point against the use of fossil fuels to generate electricity is that only one-third of the available energy is converted into electricity; two-thirds (2/3) is released as heat. Heat is dissipated in cooling towers or by heating river or sea water with the associated ecological consequences.
RED has none of these mentioned disadvantages. First of all, rivers will continue to flow as long as the sun shines (5 billion years according to astronomers). We then speak of renewable, sustainable energy. Secondly, the only 'reaction product' formed in RED is brackish water. This clean return water is returned to the sea and consists of half seawater that already came from the sea and half fresh water that would otherwise have flowed to the sea.
Then the issue of thermal contamination. If we manage to extract electrical energy from the mixing process, then that energy has to come from somewhere. This can only be done by cooling the mixture. The effect is small, less than 0.1 degree Celsius, but there is certainly no thermal pollution in energy extraction with RED.
The added value of RED
Ten opzichte van andere energiebronnen heeft RED een aantal specifieke voordelen.
RED is inexhaustible (sustainable) and environmentally friendly. As long as rivers flow into the sea, this technique can be applied. There are no by-products, no CO2 production (no fossil burning) and no warming of surface water takes place.
RED can be applied on both a small and large scale. Many technological processes have a scale factor that makes large-scale deployment much more profitable. However, scale is expected to play less of a role in RED. This means that the use of RED can also be a real option in remote locations for small communities.
RED is independent of wind or sunshine. RED depends only on variations in river flow that are usually reasonably predictable. However, a RED plant can be designed to a minimum flow or to a flow that is available 95% of the time, a very high load factor
RED can be deployed at times of greatest demand when a reservoir like the IJsselmeer is present. “Fuel” is then saved during the off hours. For the IJsselmeer this only becomes interesting if practically all the incoming water is used by the RED installation. In that case the water level in the IJsselmeer would rise by 4 cm per day. A day/night system is therefore quite possible.
RED provides ecological added value in a number of applications. One example is the dilution of the concentrate (brine) produced by plants for desalinating seawater. Something similar occurs when water is discharged at the Lorentz and Stevin locks in the Afsluitdijk. For a short period of time (from about 2 hours before to 2 hours after low tide) the water can be discharged. An enormous amount of fresh water then takes possession of a large part of the area that used to be salty. Many marine animals are poorly able to withstand the resulting osmotic shock.
RED also performs a pumping function. The rising sea level will make it more difficult to discharge water from the IJsselmeer lake into the sea. The use of pumping stations is already being considered. If the Afsluitdijk were to be used, part of the energy generated would be used to pump the water from the IJsselmeer lake to the sea via the installation. A RED installation can thus become a combination of power plant and pumping station.
Stack construction
REDstack B.V. takes its name from the technology called RED (Reverse ElectroDialysis) and its practical implementation with a stack of membranes. This membrane stack consists of many hundreds of membrane pairs and the final voltage is the sum of the individual voltages. A membrane pair is made up of two different membranes: an AEM (anion exchange membrane) and a CEM (cation exchange membrane) that with spacers in between, tissues with a fairly open structure through which fresh (river water) and salt (seawater) flows. The salt in the seawater wants to diffuse into the river water. In doing so, the positive Na+ ions will only be able to pass the CEM and the negative Cl-- ions will only be able to pass the AEM. Positive and negative ions thus move in opposite directions in the stack. On both extreme sides of the membrane stack are the electrodes where, via a redox reaction, the ionic current changes into an electric current.
Construction
The membrane stack is placed in an enclosure that provides the supply of fresh and salt water and the drainage of brackish water. In addition, the enclosure houses the electrodes. The stacks for laboratory experiments have been further developed into stacks used at the REDstack test and development site on the Afsluitdijk. The aim is to optimize the stacks: the goal is maximum power generation and a long lifetime, coupled with minimum pollution. At the same time, REDstack is gradually scaling up the electrical power supplied by these stacks by increasing the membrane area and improving the flow, as well as reducing production costs.
Development
REDstack BV is engaged in the production and development of stacks specifically for RED and ED technology. Not only the production of the membrane stack, but also development of the housing around the stack is part of REDstack's expertise. For the project on the Afsluitdijk there is a close collaboration with membrane producers and with Wetsus. Wetsus specializes in pollution control where the old saying applies: 'prevention is better than cure'. In practice, this means good coordination of the stack design and the method of pre-purification of sea and river water.